Surgeon-Specific Information

Clinically Proven Acromioclavicular Joint (ACJ) reconstruction

Lockdown Shoulder Stabilisation System (LSSS) is a synthetic device used in acromioclavicular reconstruction to replace the ligament and recreate the anatomy. The device is made of double braided polyester, with a tensile strength of 1730N. There are over 3,000 implantations being made every year worldwide. LockDown allows early mobilisation at 2 weeks, quick rehabilitation – with light exercise at 8 weeks and contact sport at 12 weeks.

Surgical Technique

In the event of an acromioclavicular joint (ACJ) dislocation, LockDown provides an elegant and easy solution: a synthetic braid that loops around the coracoid, passes around the posterior of the clavicle and then is secured with a single screw in the anterior-posterior direction. This provides stability while allowing natural rotation of the clavicle.
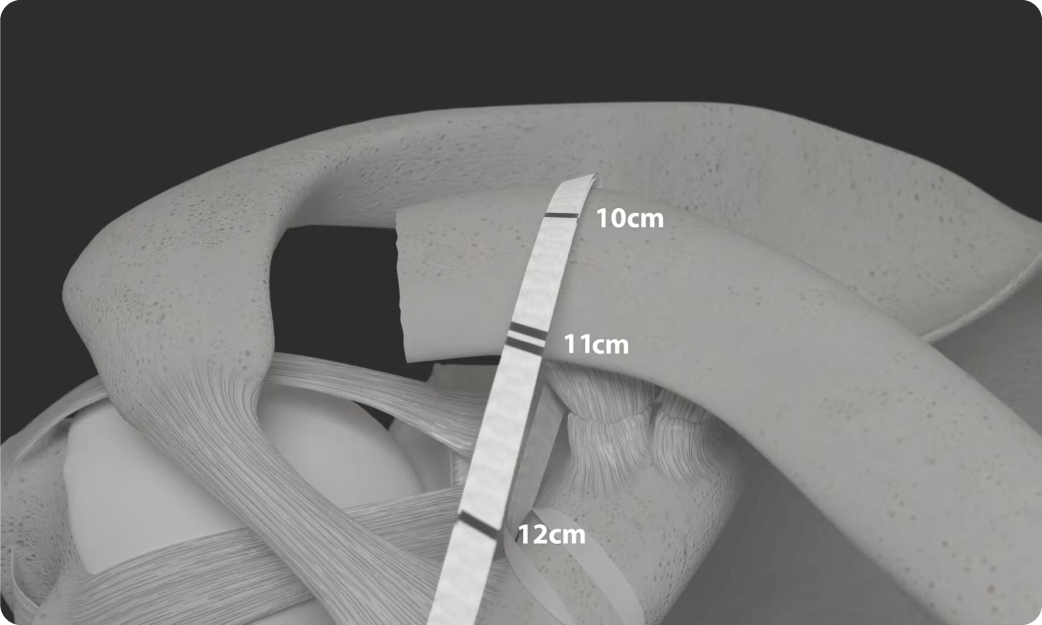
LockDown is an open procedure and has been design to augment reconstruction of the acromioclavicular joint ligaments as a result of separation through injury. The device is available in a variety of lengths to accommodate the differing anatomical features in each patient.
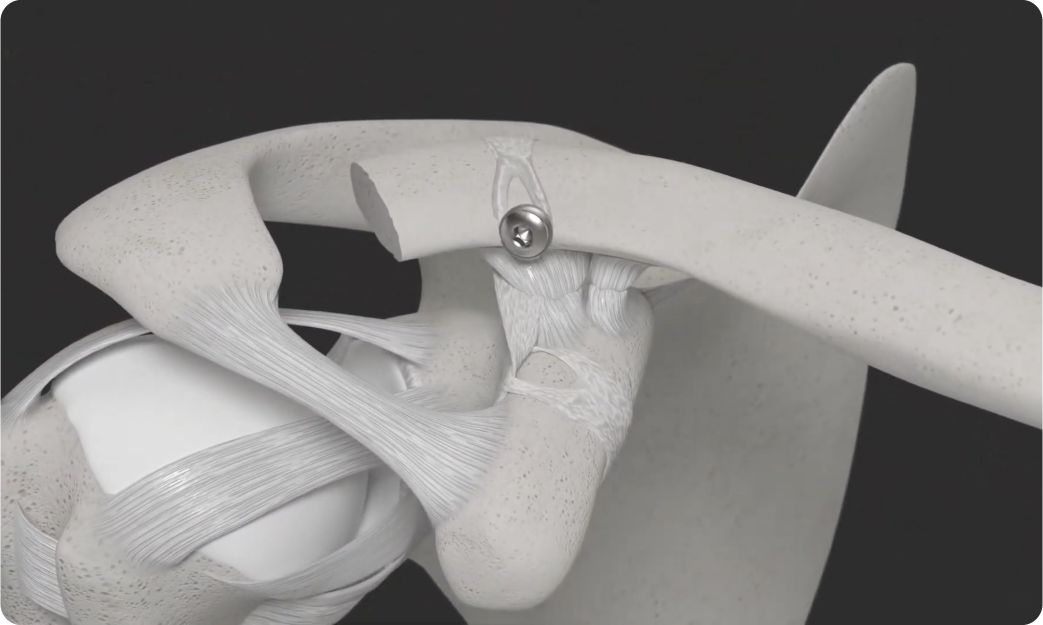
LockDown is luggage tagged around the coracoid process, requiring no large holes, it is passed behind the clavicle and fixed with a small screw and washer to the front of the clavicle, allowing the clavicle to rotate.
Indications
- Acromioclavicular dislocation (acute & chronic)
- Rockwood Type III, IV & V acromioclavicular joint injury
- Lateral clavicle fractures
- Revision of a previous stabilisation procedure such as a failed Weaver Dunn

- LockDown LSSS for ACJ reconstruction, was originally launched as ‘Surgilig’ in 2004 and part of a company dating back to 1924 with an impressive pedigree in innovation borne out of collaboration with clinicians. It was developed by Prof. Angus Wallace and team in Nottingham University Hospital. The name was changed from ‘Surgilig’ to ‘LockDown’ in 2011, as part of FDA approval process.
- Over 40,000 LockDown Ligaments have been implanted to date worldwide.
- The system boasts more than 20 years of clinical evidence, combined with exceptional patient outcomes, backed up by worldwide clinical use.
- Straightforward reproducible surgical technique with simple instrumentation.
- Used for acute and chronic AC injuries. Grade III, IV and V Rockwood score.
DOWNLOAD CLINICAL STUDY COMPENDIUM
The system boasts more than 20 years of clinical evidence, combined with exceptional patient outcomes, backed up by worldwide clinical use, with thousands of the device implanted each year.
Clinical Studies detailed in the Compendium include:
- Chronic acromioclavicular separation: The medium term results of coracoclavicular ligament reconstruction using braided polyester prosthetic ligament
- Preliminary Results of the ‘SurgiligTM’ [Lockdown LSSS] Synthetic Ligament in the Management of Chronic Acromioclavicular Joint Disruption
- Stabilization of acromioclavicular joint dislocation using the ‘Surgilig’ [Lockdown LSSS] technique
- Acromioclavicular joint dislocation: diagnosis and management
- Modified Weaver-Dunn procedure versus the use of a synthetic ligament for acromioclavicular joint reconstruction
- The biological response to a failed extra-articular polyester ligament used for AC Joint reconstruction at the shoulder girdle
- Stabilisation for the disrupted acromioclavicular joint using a braided polyester prosthetic ligament
- Acromioclavicular joint reconstruction using the LockDown synthetic implant
- Current practice in the management of Rockwood type III acromioclavicular joint Dislocations
- Operative Versus Non-operative Treatment of Grade III Acromioclavicular Joint Dislocations and the use of SurgiLigTM [Lockdown LSSS]
- Anatomic reconstruction of acromioclavicular joint dislocations using allograft and synthetic ligament
- New possibilities: the LockDown device for distal clavicle fractures
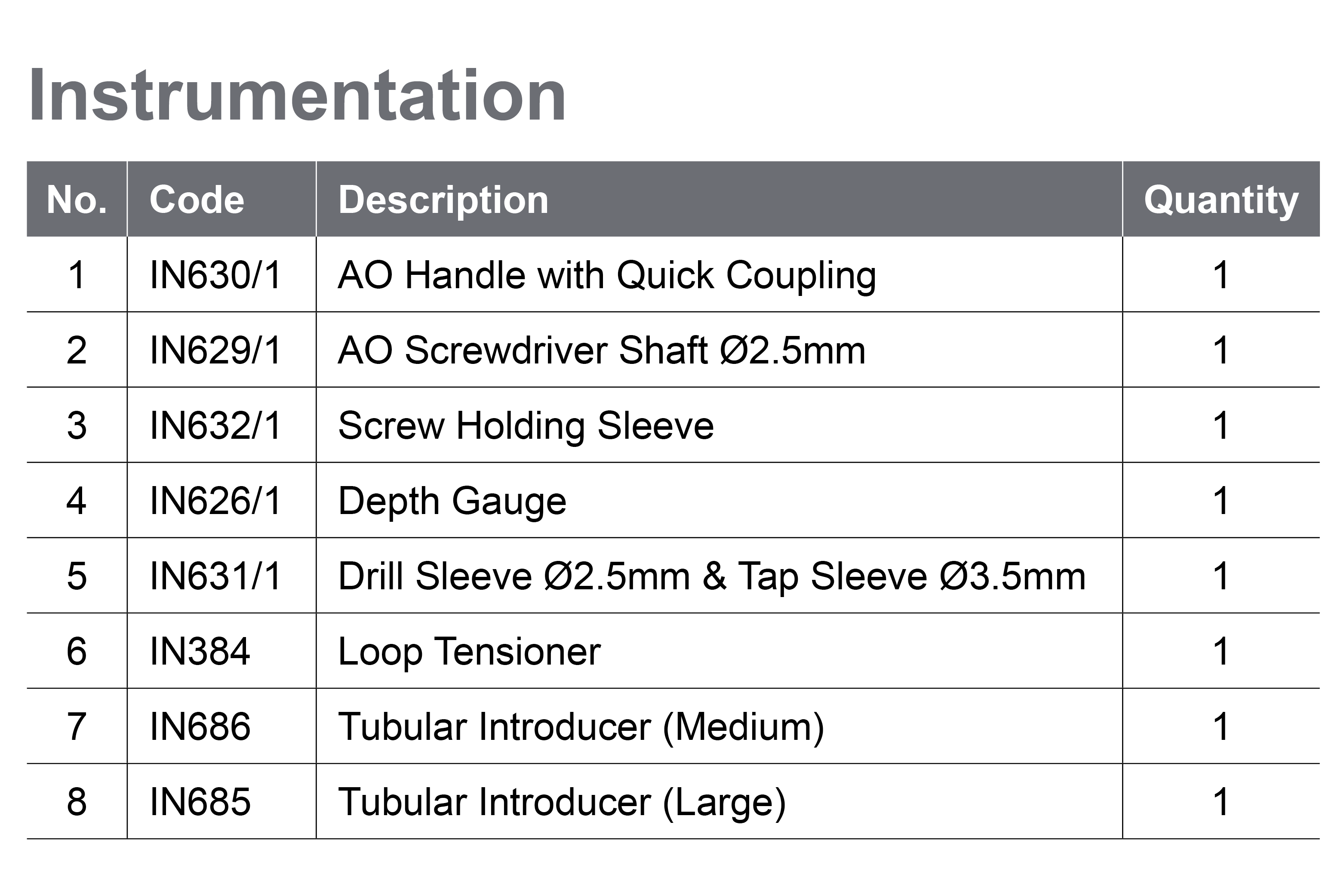
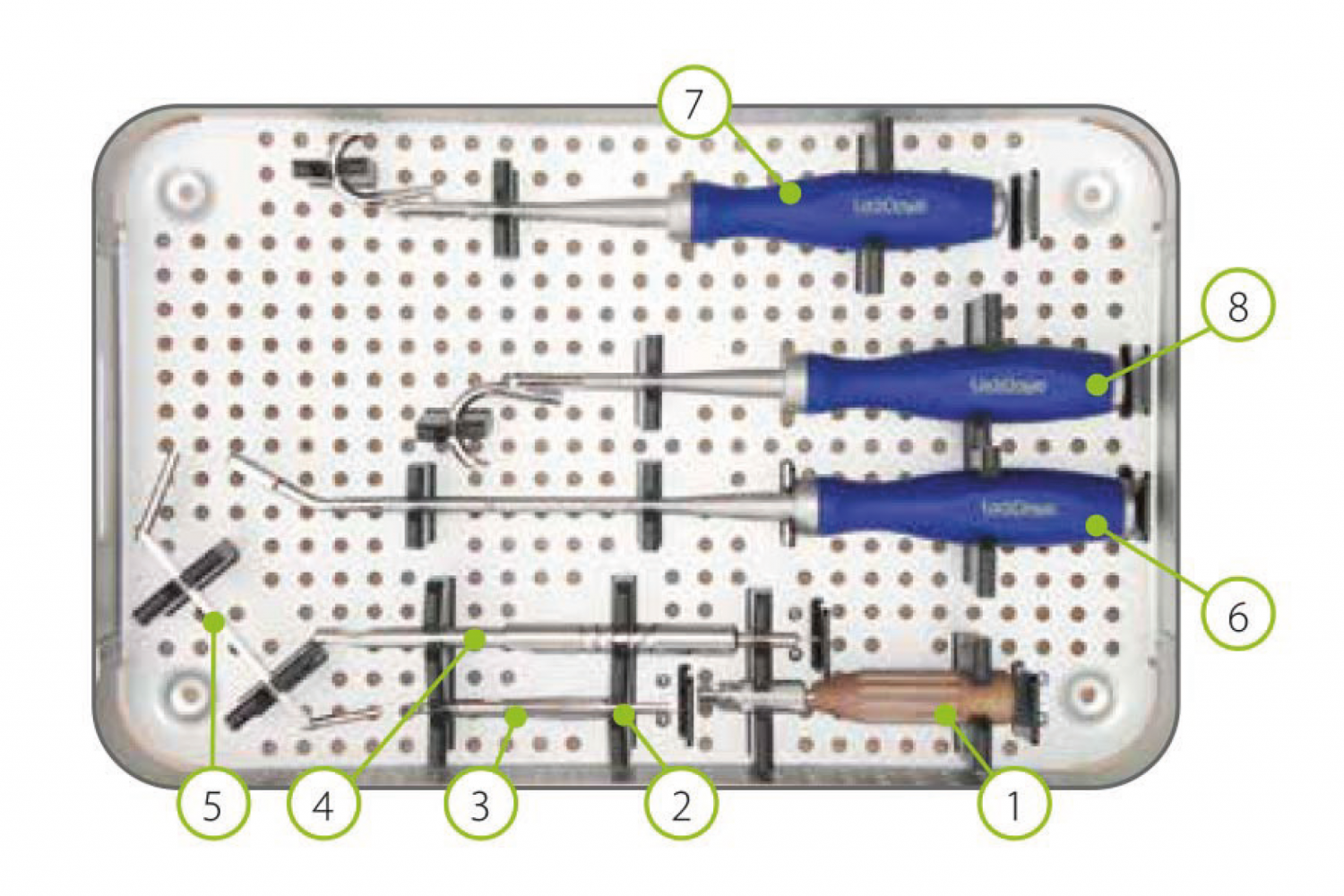
To learn more about LockDown, please visit our manufacturer partner website here
CONTACT US TODAY


